Ask An Expert
Frequently Asked Questions
Yes, We can supply simple stand alone panels or automated PLC controlled systems. We normally install and test all controls on our mixers before they are shipped.
Yes, we normally test the mixers before they are shipped and mark out the wire need to connect on the control box.
We manufacture specialty mixing equipment for powder & bulk materials. Included are ribbon blender, plough mixer, conical screw mixer, twin shaft paddle mixer, V blender, double cone blender and other auxiliary equipment such as screw conveyor, quantitive auger filler.
We sell across the world, our cusotmers distribute 5 continents.
Share Us With Your Network
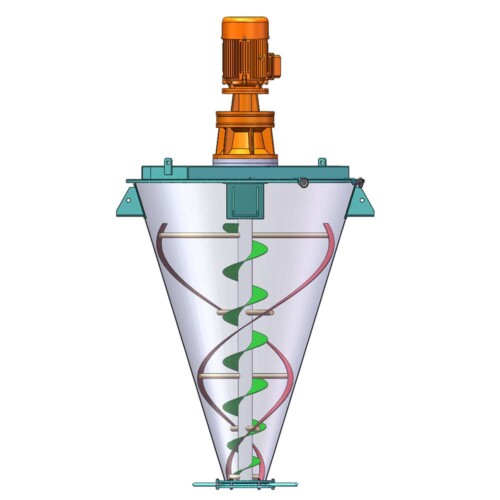
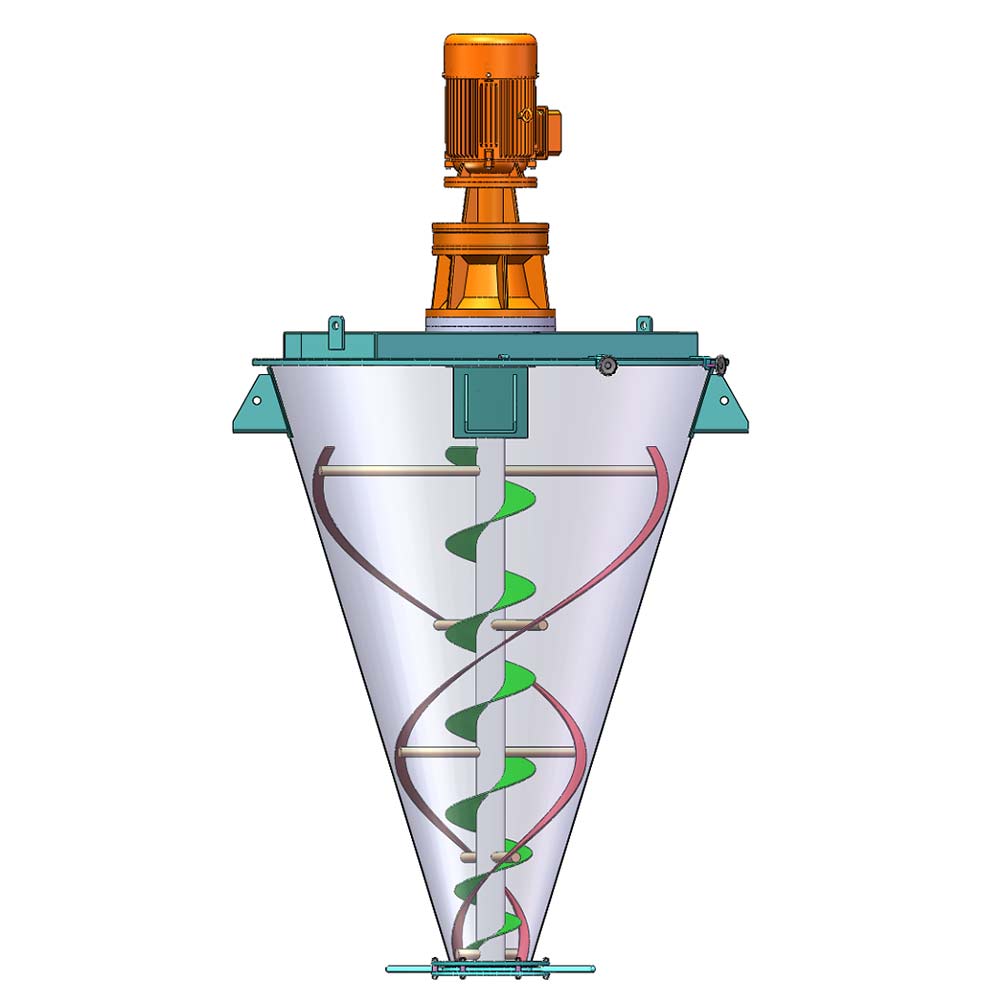
VORTEX PHARMACEUTICAL POWDER MIXING SOLUTION
Over recent decades the pharmaceutical processing has undergone a rapid transition from being a “processing art” to “processing science”. This has been possible due to increasing understanding of processing parameters, better manufacturing equipment and stricter regulatory requirements. Optimum mixing is a prerequisite for manufacturing of all solid dosage forms which involves powder mixing and it has a critical contribution in achieving uniformity of content. An understanding of powder characteristics and behavior is essential to control these operations.
Mixing is defined as shuffling type unit operation process involving both large and small particle groups and even individual particles. Mixing is energy consuming process which produces a random distribution of particles. It is dependent on the probability that an event happens in a given time and once the desired mixing has been attained, it is essential that the particles in the mix cease movement so that the system may exist in a state of static equilibrium without segregation. Some of the parameters affecting efficient mixing are:
- Particle parameters like particle size, particle shape, size distribution, particle density, cohesivity, hygroscopicity and hardness.
- Type of mixer: Speed, time, batch volume and movement
- Segregation tendency of individual components based on density difference
- Suitable mixer: Plough Shear Mixer, Ribbon Mixer, Conical Screw Mixer, Vertical Ribbon Mixer.
This review explores the nature of the mixture formulations and tries to draw conclusions for the selection of a suitable mixer for the manufacture of quality and stable products.
DRY MIXING:
Mixing of solid powders has gradually evolved from being an empirical process to being a meticulously controlled process. Much of the scientific understanding of mixing process has emerged from the behavior of non-cohesive binary particulate systems. For mixing to occur the individual particles must be redistributed repeatedly within the bulk, either by tumbling, shearing, scooping, kneading or impaction. When the powder particles are freely distributed within the bulk, there will be formation of either free flowing or non-free flowing (cohesive) mixtures depending on the particle size. Particles of larger diameter tend to be free flowing and particles of small diameter tend to be cohesive due to inter particulate forces associated with the individual particles. Significant strides have been made in understanding the dry mixing process. This includes use of powder flow analysers and computers. Recent studies have also established the use of Near Infra Red Spectroscopy (NIRS) and UV- Spectroscopy which can be utilized to ensure the homogeneity of powder mixture. Nowadays, Plough Shear Mixer is most popularly used in pharmaceutical industry which can make high homogeneity within short mixing time.
TYPES OF MIXTURES:
- Free flowing mixtures
Free flowing powders have desirable features like minimal need of lubricant and effective contact with die cavity. They suffer from a serious drawback of segregation of individual components in post mixing processing. The particles can move smoothly and independently in a particular direction due to the interparticulate forces. The free flowing powders need to be handled and stored in a proper manner by packing the products in polythene bags and applying vacuum, before sealing.
- Cohesive mixtures
The cohesive mixture exhibits “stick-slip” characteristics and the components are not free flowing, the individual particles are repeatedly broken down and allowed to redistribute within the system to ensure a satisfactorily mixed product. The scale of segregation is less, but the intensity of segregation will be more due to the retaining of structure by the small agglomerates throughout the mixing process. Some of the parameters contributing to the formation of cohesive mixtures are moisture, electrostatic charges and solid bridges between the particles.
- Ordered mixtures
If one of the constituents of the powder mix is added to a fine, micronized form then on mixing the larger particles may adsorb some of these smaller particles to active sites on their surface where they are held tenaciously. Ordered mixtures are formed by mechanical, adhesion or coating forces such that the ordered unit will be the smallest possible sample of the mix and will be of near identical compositions to all other ordered units in the mix.
MECHANISM OF MIXING:
Mixing involves the following steps
- Convective movement of relatively large portions of the bed (Macromixing)
- Shear failure, which reduces the scale of segregation
- Diffusive movement of individual particles (Micromixing)
A quantitative theory was developed to describe mechanism of mixing in particulate systems in which one component is present only in a trace quantity. Theoretical expressions were derived to predict the role of mixing of the drug moiety.
MIXER SELECTION:
An ideal mixer should be capable of producing complete blend in reasonable time without damaging the product. Additionally it should be dust-tight, require low maintenance and energy, can be discharged and cleaned easily. All these properties cannot be found in a single mixer. However, a logical selection of a mixer can be done based on
- Powder characteristics of the constituents of the mixture
- Quality requirements of the product
- Process requirements and limitations.
Additionally the following points need to be considered, to ensure overall success of a mixing process
- Does the mixer have flexibility to cope with a variable batch size?
- Can the mixing vessel be transported between the operations like loading, mixing, packaging?
- Does it have easy access for sampling?
- How well can the mixer separate the process materials?
- Does the mixer require frequent cleaning? If so what are the standards?
- What should be the nature of the mixing surface?
The effectiveness of a mixer depends on the powders to be mixed, the time of mixing, number of rotations of the mixer and other factors, which may be characterized by complex mathematical relationship. The performance of the mixers can be predicted by using different powders with the general theory of powder mixing using Powder mixing monitor.