Ask An Expert
Frequently Asked Questions
Yes, We can supply simple stand alone panels or automated PLC controlled systems. We normally install and test all controls on our mixers before they are shipped.
Yes, we normally test the mixers before they are shipped and mark out the wire need to connect on the control box.
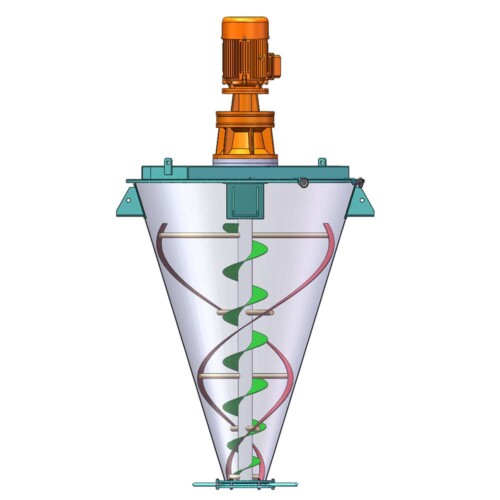
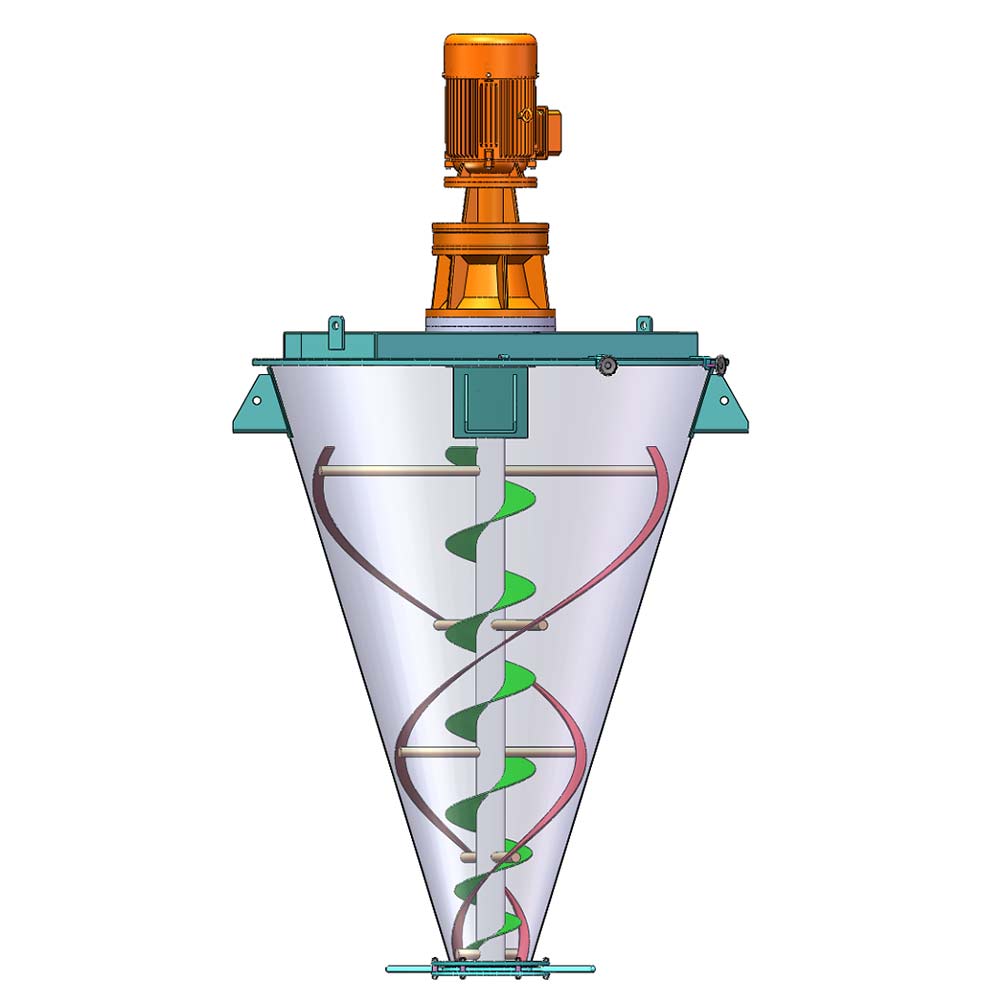
We manufacture specialty mixing equipment for powder & bulk materials. Included are ribbon blender, plough mixer, conical screw mixer, twin shaft paddle mixer, V blender, double cone blender and other auxiliary equipment such as screw conveyor, quantitive auger filler.
We sell across the world, our cusotmers distribute 5 continents.
Share Us With Your Network
Mixing Intensity versus Mixing Time
Every task requires a defined amount of energy to accomplish. This statement also embraces the science of mixing.
Mixing is, in the typical industrial context, the intermingling of ingredients to the extent that is acceptable for the final process, with acceptance being defined as suitability for the product’s final usage. Mix homogeneity greatly affects the product’s qualities or reject rate.
When mixing solids and /or liquids, the objective is to disperse the ingredients in such a way that these initially segregated ingredients are repositioned randomly in the mix. Whether we view this task as material transportation or interfacial area creation, the process takes energy to accomplish. Mixing comes from tools or medium acting on the ingredients. The motive energy originates from the drive motors driving these tools /medium or the dynamic energy within the ingredients.
Material characteristics such as particle size, density, and shape as well as cohesive and adhesive tendencies all affect the ease, alternatively difficulty to achieve a good mix. For free‐flowing ingredients of similar size and density, such as granular materials without cohesive influences, very little mixer action is required. A low intensity mixer inputting as little as 5 hp per 1000 pounds of mix could be adequate to produce a good mix within a reasonable batch cycle. A recipe consisting of largely powdery and fibrous ingredients on one hand and a tightly agglomerated minor ingredient on the other will present a much greater challenge. In such cases, the mixer is asked not only to mix the ingredients, but also to separate the fibers and de‐agglomerate the lumps. Almost intuitively, we recognize there is some minimum intensity that is required to handle these additional tasks.
The preceding description touches on some of the basic energy‐related considerations in mixing:
1. A certain minimum level of intensity is needed to achieve a good mix. That minimum level of intensity is dependent on ingredient characteristics and their interactions.
2. A fixed amount of energy is needed to disperse an equal amount of ingredients having the same characteristics to the same degree of homogeneity.
3. Mixing energy should be distributed to the batch evenly.
4. Mixing time and intensity are inversely related through mixing energy.
Therefore, it is no great revelation that generally, if specific intensity (i.e. intensity per unit weight of the mix) doubles, mixing time to the same level of dispersion can be halved.
For batch processes, one can manipulate intensity by adding / removing mixing tools, or one can increase the speed of the mixing tool, or both. The speed approach generally follows the relationship that intensity varies as the square of the tool speed. For example, doubling the peripheral speed of an agitator from 10 ft per second to 20 ft. per second increases intensity 4 fold, hence resulting in mixing time being reduced to ¼. Suddenly, the same mixer’s production capacity is almost quadrupled!
How can we be confident that the above is the case? The answer is: we can follow experimentally with the use of statistical analysis on mix samples drawn through two mixing cycles, one at a preset base tool speed, the other at, say, double that speed. A commonly used parameter called “mixing index” allows us to map the progress of dispersion.