Ask An Expert
Frequently Asked Questions
Yes, We can supply simple stand alone panels or automated PLC controlled systems. We normally install and test all controls on our mixers before they are shipped.
Yes, we normally test the mixers before they are shipped and mark out the wire need to connect on the control box.
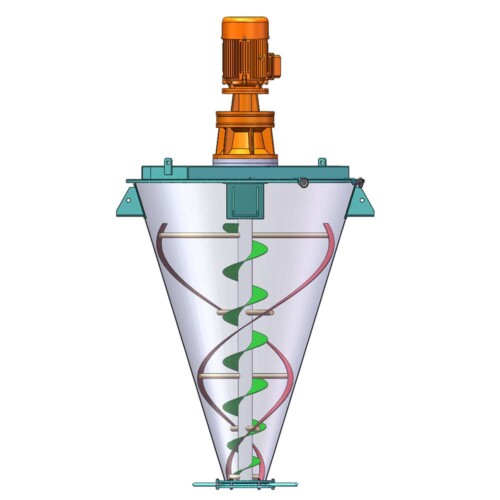
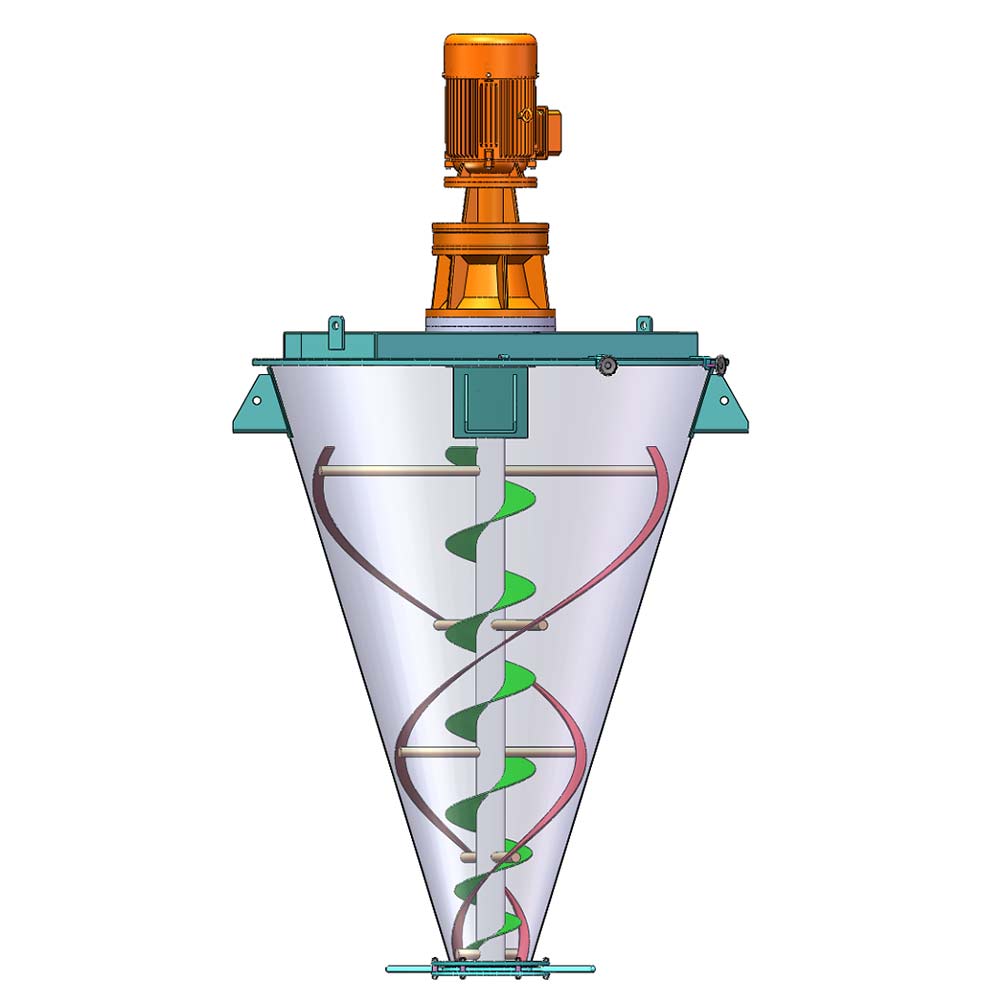
We manufacture specialty mixing equipment for powder & bulk materials. Included are ribbon blender, plough mixer, conical screw mixer, twin shaft paddle mixer, V blender, double cone blender and other auxiliary equipment such as screw conveyor, quantitive auger filler.
We sell across the world, our cusotmers distribute 5 continents.
Share Us With Your Network
Mixing and direct compression
Q: Our pharmaceutical process currently requires a granulation step. We’d like to skip this step and the processing and equipment it requires and go to direct compression. What should we consider before making this change?
A: With pharmaceuticals, using direct compression (transferring a mixture from a mixer directly to a tablet press) typically involves a mixture with a low concentration of active ingredients. This can cause the same problems as with mixing minor ingredients. For instance, if the size difference between the largest and smallest particles is 5 to 1 or greater, the mixture is likely to segregate. In some situations, direct compression can be dangerous because it makes controlling mixing quality difficult. With certain pharmaceuticals with a particle size difference 200 to 1 or greater (such as 200-micron excipients mixed with l-micron active ingredients), segregation is so significant that you may have to avoid direct compression and use granulation to maintain the final product’s integrity. In some cases, you can avoid segregation problems by reducing your material‘s particle size difference (for instance, by grinding the larger particles) to less than 5 to 1.
If you can’t reduce the particle size difference, reduce segregation problems by using few transfer points and remixing the material in the hopper or other container just prior to loading the tablet press. (The material can be remixed by using equipment designed to rotate and tumble-blend the hopper or other container.) If making these changes doesn’t eliminate segregation problems, use granulation – either wet, by adding a liquid binder, or dry, by compressing the mixture between rollers. In either case, you’ll need additional equipment, which will raise the system’s cost.